|
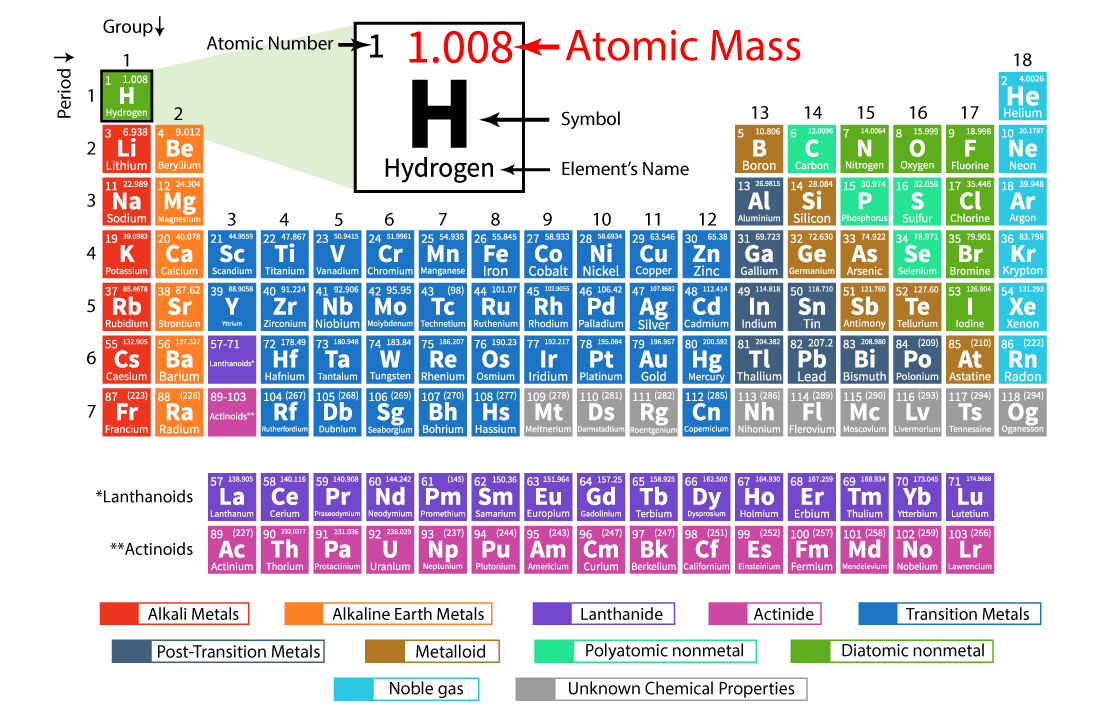
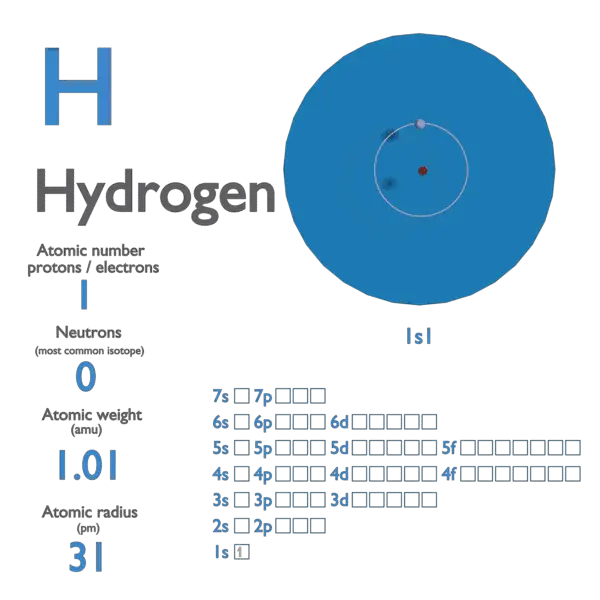
Unlike the number of protons in an element, which never changes, the number of neutrons in atoms of a certain element can vary often enough that the average atomic mass of the element must be expressed as a decimal value between two whole numbers. While 2 atoms with the same number of protons and differing numbers of neutrons are both the same element, they are different isotopes of that element. The number of neutrons can vary among atoms of a certain element. Though, especially among elements at the top of the periodic table, it may seem that an atoms' atomic mass is about twice its atomic number, atomic mass isn't ever calculated by doubling an element's atomic number.įind the number of neutrons in the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
